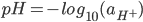Effect of pH on the Interfacial Tension of Lipid Bilayer Membrane. Aneta D. Petelska and Zbigniew A. Figaszewski. Biophysical Journal, Volume 78, Issue 2, 812-817, 1 February 2000.
A theoretical equation is derived to describe the dependence of the interfacial tension of a lipid bilayer on the pH of the aqueous solution. Interfacial tension measurements of an egg phosphatidylcholine bilayer were carried out. The experimental results agreed with those derived from the theoretical equation obtained close to the isoelectric point within a range of three pH units. A maximum corresponding to the isoelectric point appears both in the theoretical equation and in the experiment.
abstract here